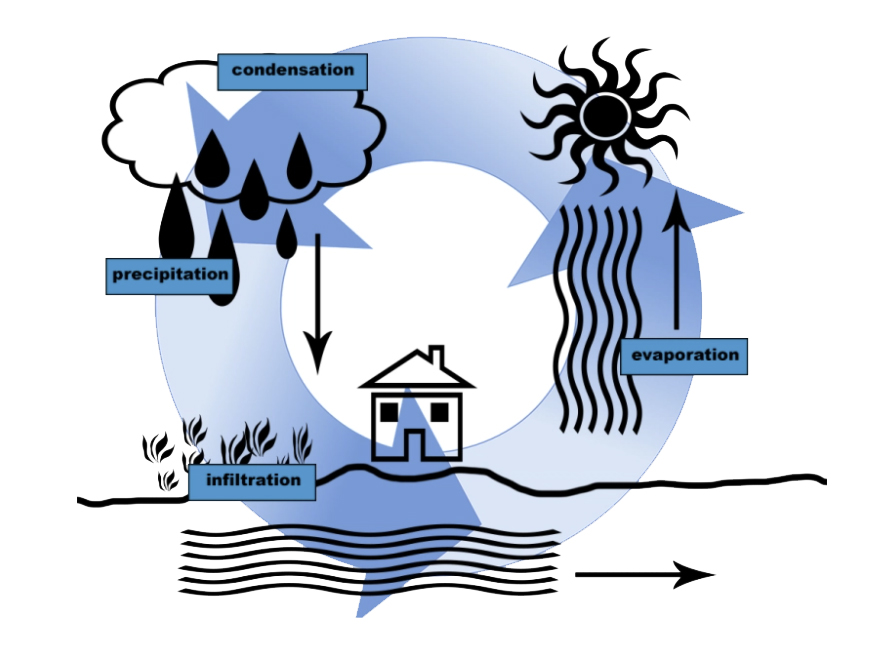
Our planet earth has always had the same amount of water. The water we use today was also there for the dinosaurs to drink. And through the water cycle, water can be found everywhere. It is always moving and changing states as a liquid, solid, or gas.
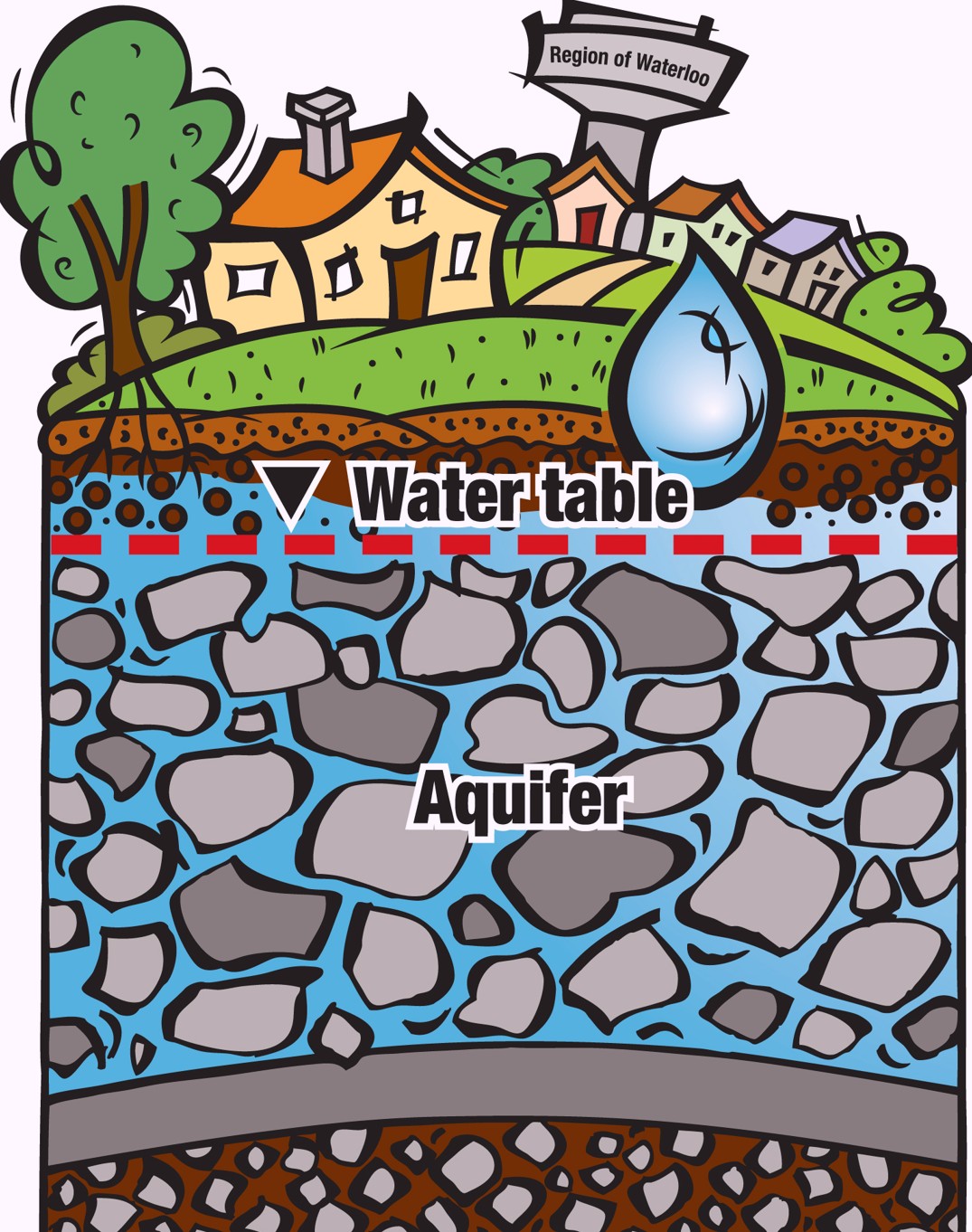
We can find water in streams, rivers, and lakes. It’s in the air and underground. It is the glaciers, rain, and snow. And it’s in our food, plants, and bodies.
Earth has a lot of water – about 71 per cent of our planet is covered in water. But how much of it is drinkable fresh water? First look at the oceans – that’s a lot of water. In fact, 97 per cent of all water is salt water – water we can’t drink without desalinating or removing the salt. That leaves three per cent remaining as fresh water. Some of the fresh water is hidden underground as groundwater. Water is always moving, even if it takes the slow route, deep underground through rocks and sand that make up the layers in an aquifer where we find groundwater.
Locally, Waterloo Region is part of the Grand River watershed. But what is a watershed? Is it a shed made out of water? Nope. A watershed is an area of land that drains into the same body of water. Think of it like a tree branch. In Waterloo Region, groundwater seeps up into small waterways such as Schneider’s Creek, Laurel Creek and Mill Creek that drain into the larger Grand River. The Grand River and many other watersheds drain into Lake Erie and eventually into the Atlantic Ocean.
Have you ever watched the National Film Board movie “Paddle to the Sea”? The movie reminds me of a watershed. It is about a child who makes a wood carving of a man in a canoe and sets it on a frozen stream waiting for the spring thaw. The movie follows the canoe as it travels from the stream to larger waterways and finally to the ocean.
The water cycle and the fact we all live in a watershed are reminders we all share the same water, and we all live downstream from someone else.
And why we all have a role to play protecting this important resource - water.




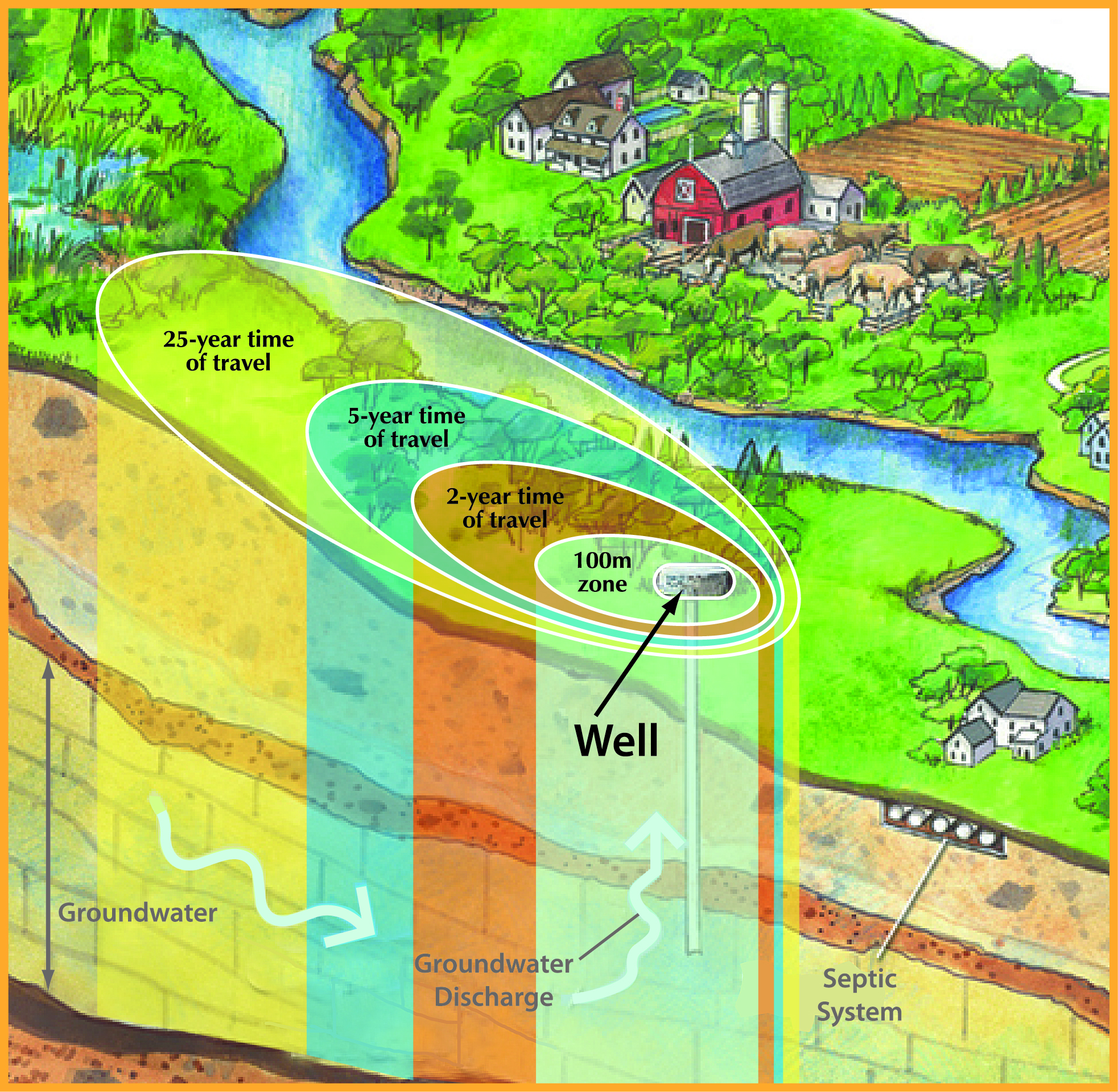 Wellhead Protection Areas surround each municipal supply well. Image credit: Conservation Ontario.
Wellhead Protection Areas surround each municipal supply well. Image credit: Conservation Ontario.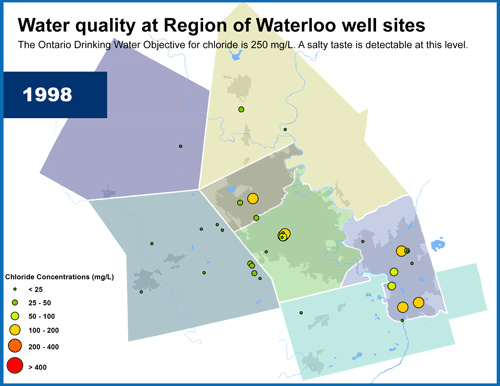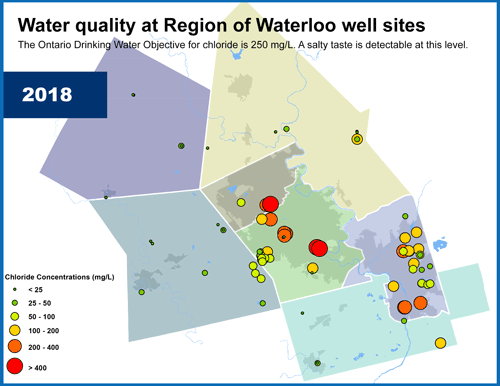



 Leaky eaves troughs and downspouts can drip water on to paved walking areas that can turn into black ice, hard to see but easy to slip on. While taking that walk on a rainy day remember to look up for drips and leaks that can freeze into ice on a walkway.
Leaky eaves troughs and downspouts can drip water on to paved walking areas that can turn into black ice, hard to see but easy to slip on. While taking that walk on a rainy day remember to look up for drips and leaks that can freeze into ice on a walkway.




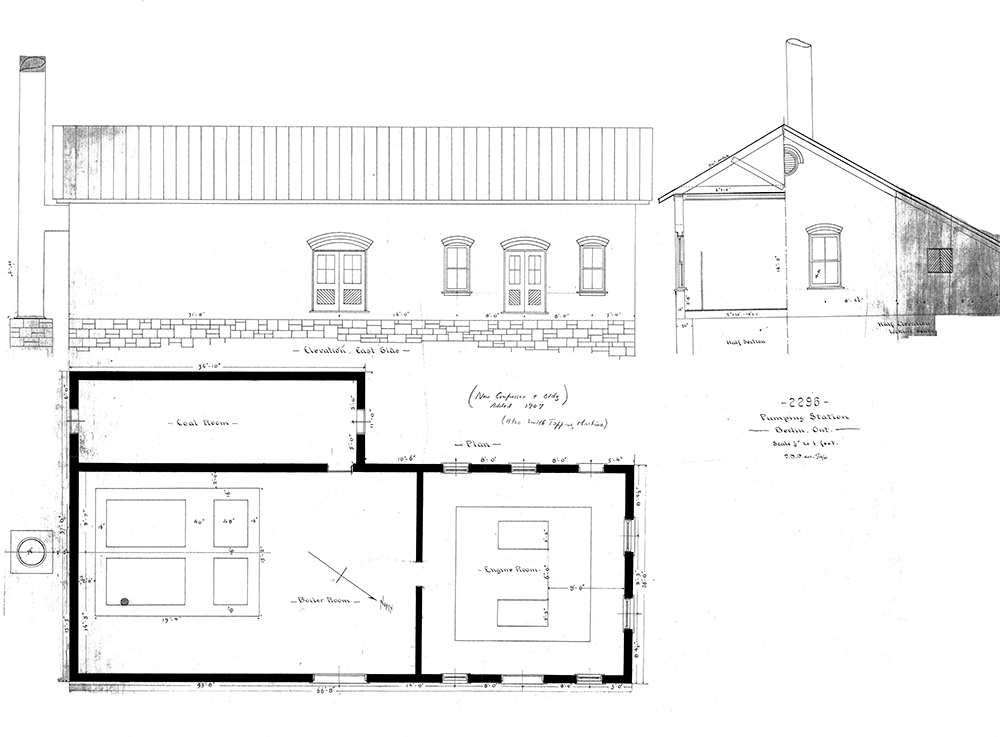
 Wood reservoir at the Middleton Water Pumping Station.
Wood reservoir at the Middleton Water Pumping Station.